Recursos e regulamentos da Cannabis na Colômbia
Colômbia aprova flor de cannabis para exportação
Atualizado: janeiro de 2023
O ambiente de cultivo na Colômbia é ideal para cultivo ao ar livre em larga escala, alavancando sua rica experiência agrícola. Com 12 horas de sol o ano todo, proporcionando múltiplas colheitas de cannabis, baixos custos de mão de obra e terra barata, a Colômbia está posicionada para ser um dos fornecedores de baixo custo do mundo.
A Colômbia legalizou a cannabis em 2016 para a exportação de óleos e extratos com baixo teor de THC e ganhou ainda mais atenção internacional quando Decreto 811 aprovou a exportação de flores secas de cannabis em julho de 2021. No entanto, foi somente em 1º de abril de 2022 que Resolução 539 quando mecanismos regulatórios reais entraram em vigor, permitindo a exportação de flores secas com altos níveis de THC.
Com essa mudança de política, a Colômbia está se posicionando para se tornar um importante fornecedor de baixo custo para o mercado global de cannabis medicinal.
Consequentemente, o investimento internacional continuou a se expandir, com o propósito de exportar para a União Europeia, Israel e Austrália. Mais de 18 multinacionais se instalaram na Colômbia.
Mercados de exportação impulsionam os padrões de qualidade da Colômbia
A União Europeia – com uma suboferta crônica de cultivadores locais – é indiscutivelmente a maior oportunidade de exportação para cannabis medicinal – e a Colômbia. À medida que o mercado evolui, os administradores europeus estão buscando uma estrutura regulatória modelada na indústria farmacêutica.
Os produtores colombianos que buscam exportar para os 27 países da UE e seus 350 milhões de residentes devem seguir as diretrizes GACP (Boas Práticas Agrícolas e de Coleta) e GMP (Boas Práticas de Fabricação) da UE para cultivo e fabricação, respectivamente, bem como os limites microbianos estabelecidos pela Farmacopeia Europeia.
E aqui o quadro começa a ficar obscuro. A Farmacopeia Europeia existente nunca teve a intenção de abordar todas as formulações e características da cannabis medicinal. Além disso, não há monografia da UE para cannabis. Consequentemente, há uma ampla gama de interpretações sobre quais Padrões Ph. Eur. devem ser seguidos para exportação para a UE quando se trata de conformidade microbiana.
A tabela abaixo resume a gama de requisitos microbianos em 5.1.4 e 5.1.8, os dois padrões mais frequentemente citados para conformidade microbiana.
A eficácia da radiofrequência é uma solução eficiente para atender aos rigorosos padrões da Ph. Eur.
| Ph. Eur. 5.1.4.1 | Ph. Eur. 5.1.4.2 | Ph. Eur. 5.1.8 – Tabela B | Ph. Eur. 5.1.8 – Tabela C |
|---|---|---|---|
| Critérios de aceitação para qualidade microbiológica de formas farmacêuticas não estéreis | Critérios de aceitação para qualidade microbiológica de substâncias não estéreis para uso farmacêutico | Produtos fitoterápicos com ou sem recipientes, onde o método de processamento (pré-tratamento) reduziu os níveis de organismos | Produtos fitoterápicos com ou sem recipientes, onde o método de processamento (pré-tratamento) não pode reduzir o nível do organismo ao requisito da tabela B |
| TAMC <100 UFC/g TYMC <10 UFC/g BTGN BTGN não detectado em 1g E. coli patogênica não detectada /g Salmonella não detectada /g S. aureus não detectada /g P. aeruginosa não detectada /g | TAMC <1.000 UFC/g TYMC <100 UFC/g BTGN não detectado em 1g E. coli patogênica não detectada /g Salmonella não detectada /g S. aureus não detectado /g P. aeruginosa não detectado /g | TAMC <10.000 UFC/g TYMC <100 UFC/g BTGN <100 UFC/g E. coli patogênica não detectada /g Salmonella não detectada /g | TAMC <100.000 UFC/g TYMC <10.000 UFC/g BTGN <10.000 UFC/g E. coli patogênica não detectada /g Salmonella não detectada /g |
A ausência de um regime regulamentar harmonioso na UE é detalhada extensivamente pelo especialista regulamentar alemão Markus Veit “Requisitos de qualidade para a Cannabis medicinal na União Europeia – Status Quo” Março de 2022.
Requisitos de importação alemães favorecem a radiofrequência
A Alemanha especificamente complicou ainda mais os assuntos para os exportadores, exigindo o registro de todas as cepas remediadas com radiação ionizante (gama, feixe de e-, raio-x) antes da distribuição na Alemanha. O registro leva de 12 a 18 meses e uma taxa administrativa de € 5.000 – por cepa. Como as cepas estão continuamente evoluindo para atender à demanda do mercado, esta é uma barreira custosa para fazer negócios na Alemanha.
O seguinte trecho de Novacana GmbH, um importador alemão, resume a situação:
“O regime regulamentar a supervisão da distribuição de cannabis medicinal na Alemanha está em constante mudança. As flores de cannabis são geralmente tratadas com radiação ionizante para protegê-las permanentemente contra bactérias e mofo e reduzir a contagem de germes. No entanto, de acordo com o Medicines Act (AMG), as farmácias são proibidas de vender produtos medicinais que tenham passado por radiação ionizante, a menos que tenham sido aprovados de acordo com o Portaria sobre medicamentos radioativos e medicamentos tratados com radiação ionizante (Designação alemã: AMRadV, Decreto de Irradiação)
Desde o final de 2019, isso estava sendo eventualmente imposto às flores de cannabis importadas da Holanda e do Canadá para a Alemanha também. As flores de Cannabis Medicinal cultivadas sob um regime EU-GMP são rotineiramente irradiadas com ondas gama para reduzir a contagem de bactérias e fungos e aumentar o prazo de validade. A menos que a distribuição esteja em conformidade com o Decreto de Irradiação (AMRadV), a lei alemã proíbe a distribuição de produtos irradiados. Este regulamento estipula que o distribuidor deve ter tal permissão ao fornecer esses produtos a outros atacadistas ou farmácias.
A submissão para esta licença é extensa e necessita de informações elaboradas sobre o processo de fabricação, equipamento de validação, bem como avaliação de risco para dados de estabilidade do produto irradiado.”
A radiofrequência é uma solução ideal para o requisito AMRadV, pois a RF é um processo térmico não ionizante e, portanto, fornece um caminho simplificado para exportação para a Alemanha, reduzindo tempo, dinheiro e despesas administrativas.
Caso de negócios para uso de radiofrequência
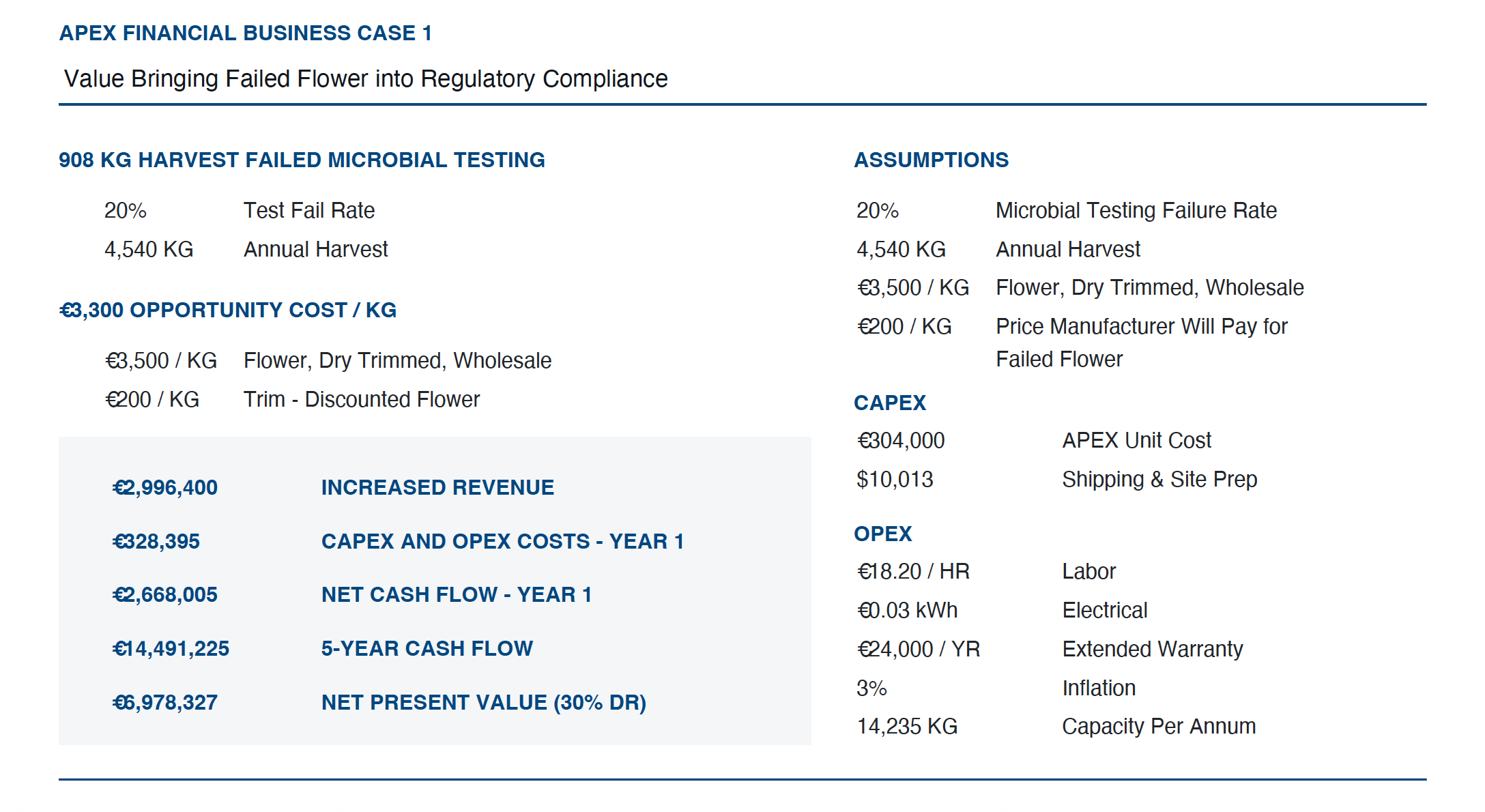
De Ziel Caso de negócios APEX permite que os clientes insiram suas próprias suposições e determinem quanto dinheiro o APEX 7 economizará para seus negócios desde o primeiro dia de remediação. O modelo analisa a porcentagem de produtos que estão falhando atualmente e traz isso para a conformidade regulatória, em vez de ter que descontar o produto com falha em até 90% e vender como acabamento para um fabricante.
No exemplo abaixo, usamos um preço de atacado de $1.200 com o produto com falha descontado para $100 e vendido como aparado. O instantâneo abaixo mostra a economia que um cultivador acumulará no primeiro ano usando o APEX 7, com base na recuperação de 20% da colheita que falha no teste microbiano em 10.000 libras de flor seca colhida por ano.
O APEX também tem o maior rendimento de qualquer tecnologia no mercado hoje, permitindo que você dimensione suas operações comerciais.
A tecnologia de cannabis limpa é uma salvaguarda vital, pois as regulamentações estão se tornando cada vez mais rigorosas. Garantir que sua colheita passe no teste microbiano aumenta sua receita bruta e otimiza seus retornos financeiros, melhorando seus resultados financeiros. Incorporar a remediação em seus SOPs pós-colheita simplifica as operações e melhora o fluxo do processo, evitando novos testes dispendiosos e faz com que seu produto chegue mais rápido aos clientes.
Sobre Ziel
A Ziel é uma desenvolvedora líder de Radiofrequência (“RF”) soluções para a redução de patógenos microbianos. As indústrias de alimentos e cannabis na América do Norte, Europa, América do Sul e Austrália contam com a tecnologia RF para remediar com segurança produtos destinados ao consumo ou ingestão humana. Os dispositivos da Ziel utilizam radiação não ionizante para pasteurizar produtos como amêndoas, castanhas de caju, macadâmias, gergelim e chia.
A tecnologia RF foi adaptada para a indústria da cannabis para remediar com sucesso patógenos bacterianos e fúngicos. Esses dispositivos ajudam os cultivadores de cannabis a garantir que estão fornecendo um produto seguro que atende aos mais altos padrões de segurança e qualidade. Além disso, a tecnologia da Ziel permite que os licenciados satisfaçam esses padrões por meio de um método compatível com os requisitos para certificação orgânica.
Contato Ziel para saber mais sobre nosso inovador sistema de processamento de cannabis para produtores sediados na Colômbia.